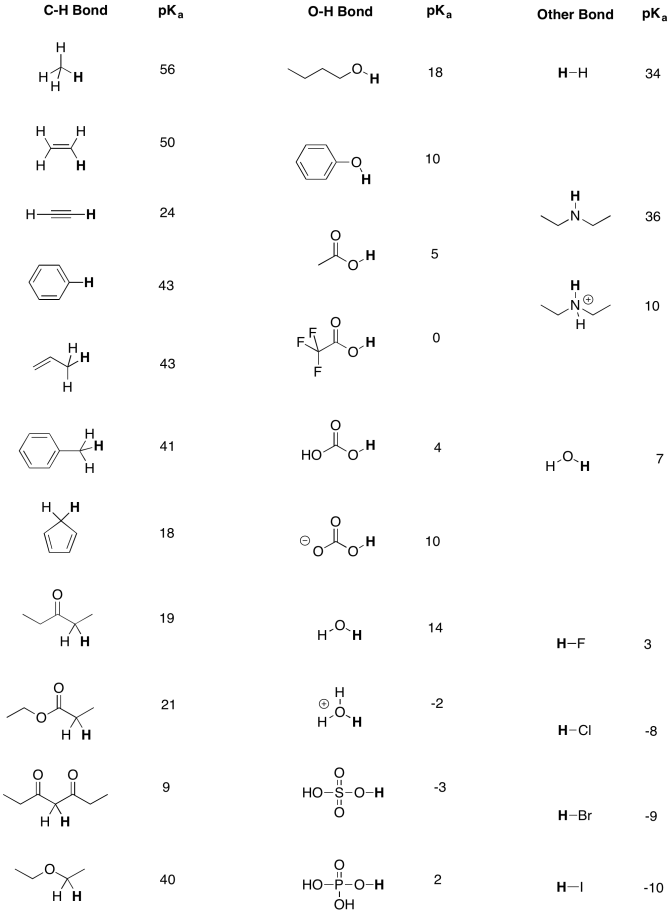
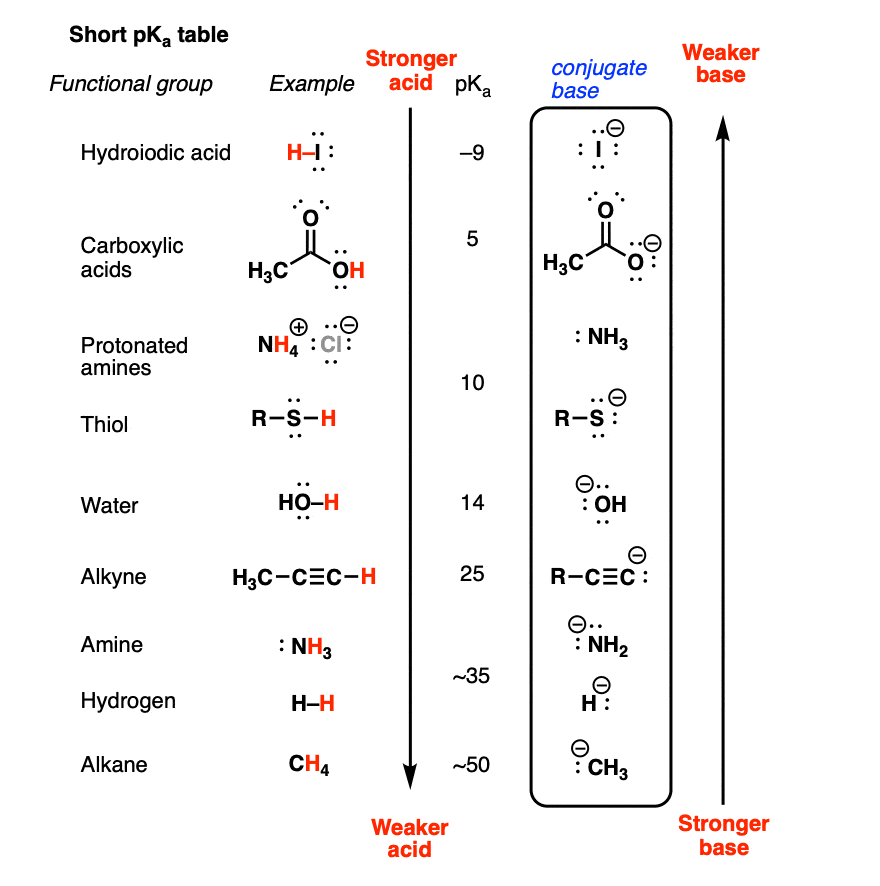
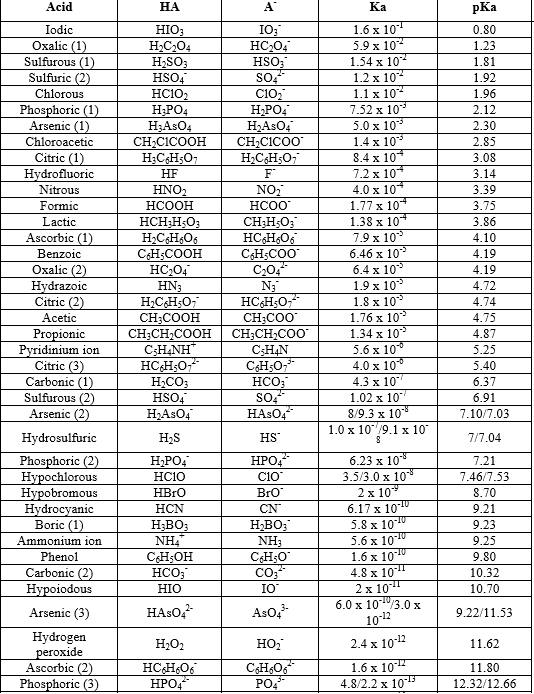
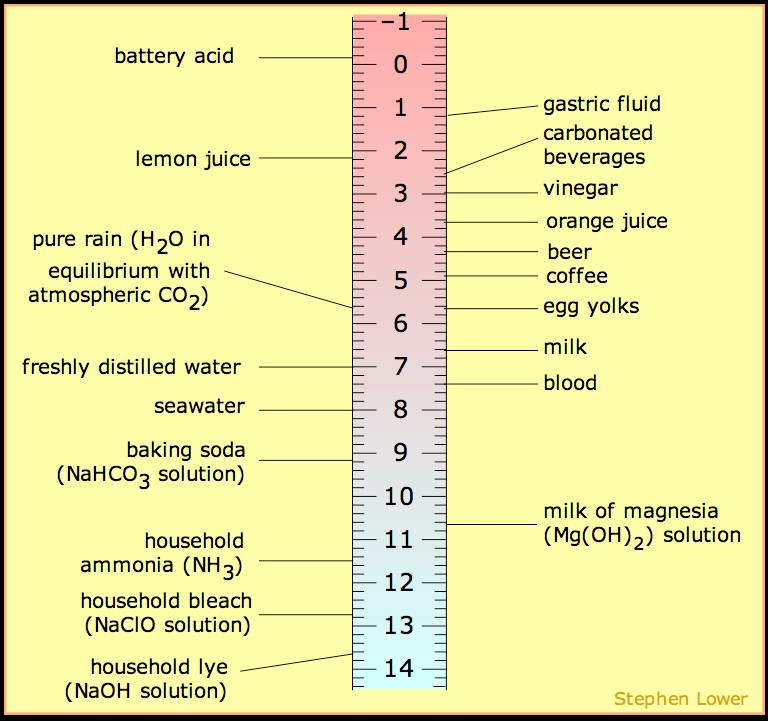
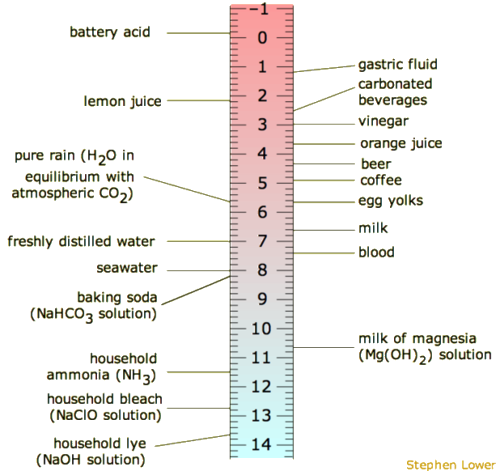
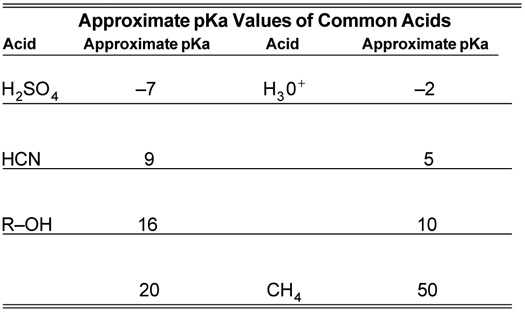
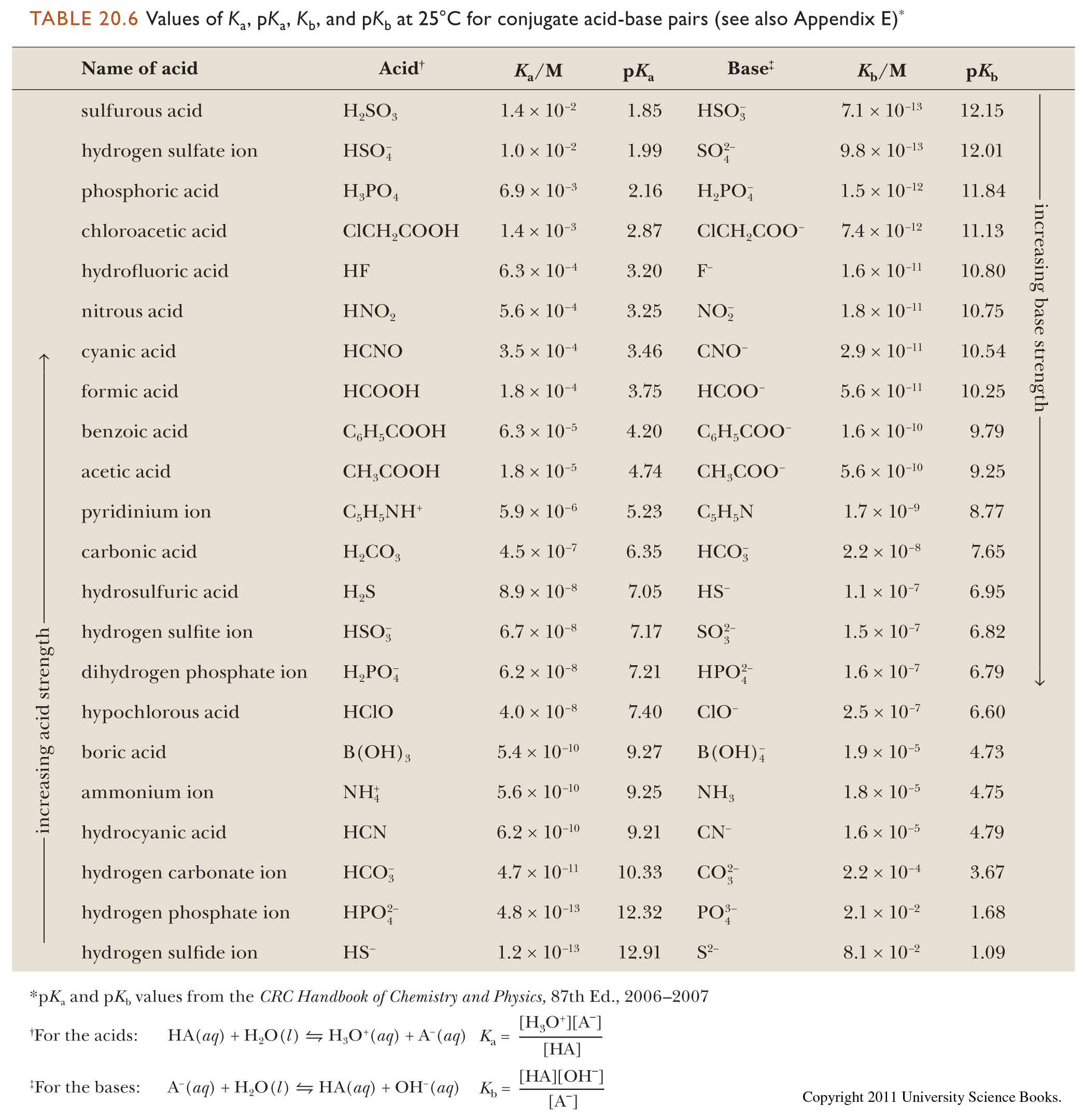
Development of Methods for the Determination of pKa Values - Jetse Reijenga, Arno van Hoof, Antonie van Loon, Bram Teunissen, 2013
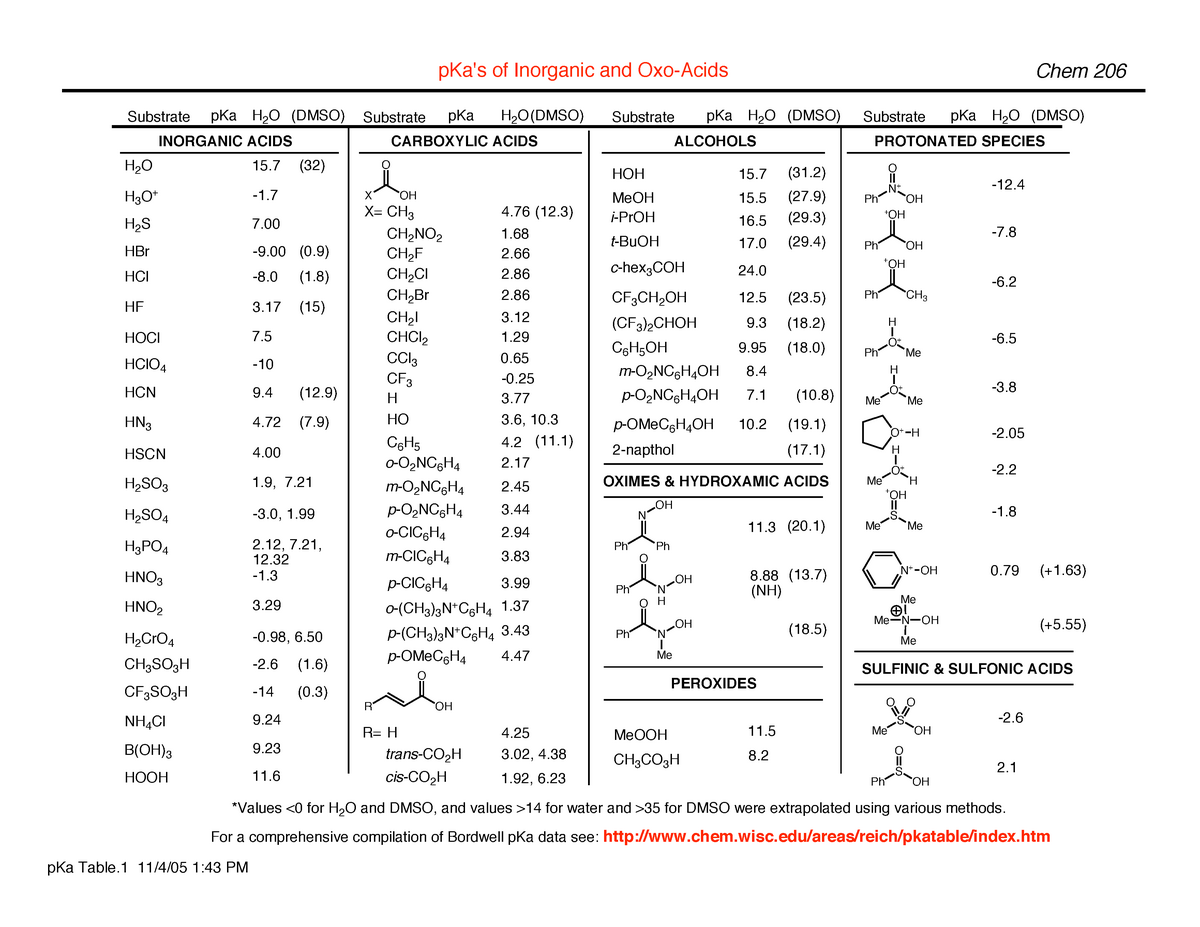
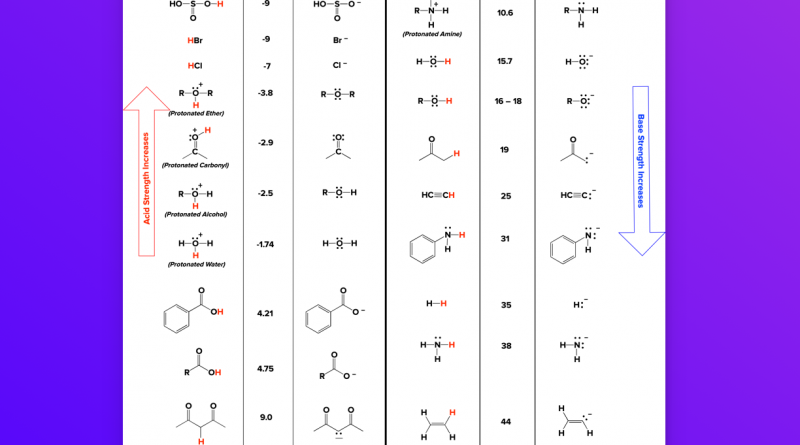
Evans pka tables - of Inorganic and Substrate pKa H2O (DMSO) INORGANIC ACIDS (32) 15 H2S 7 HBr (0) - Studocu

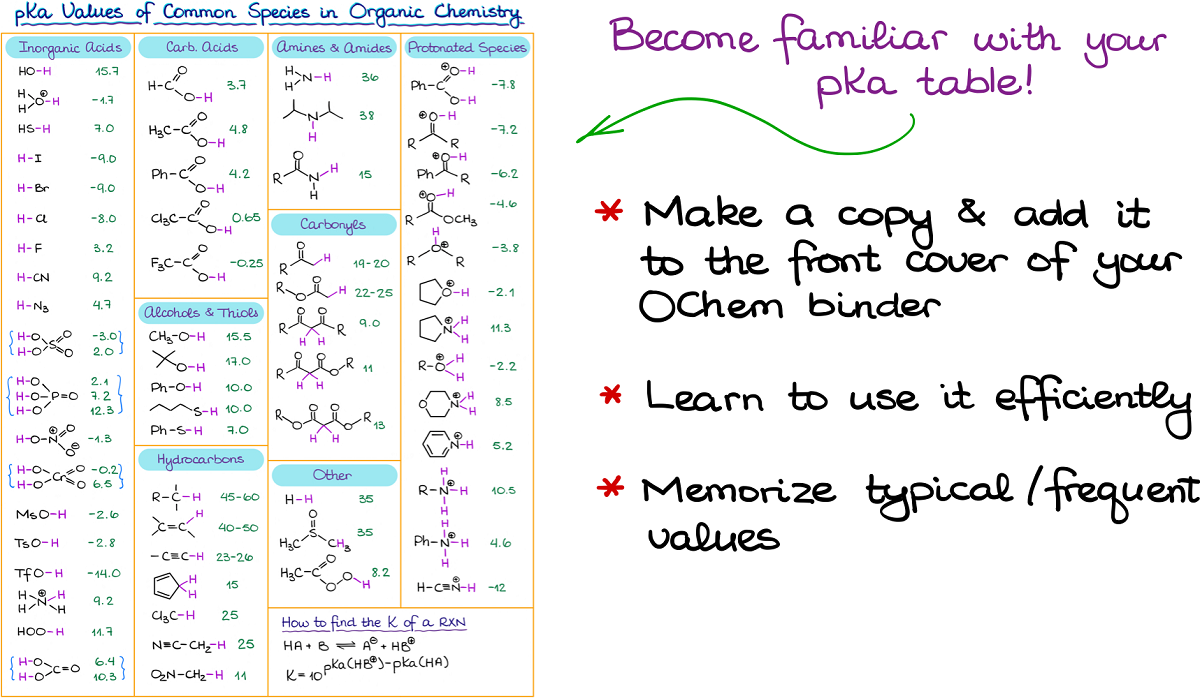
pKa & pH Values| Functional Groups, Acidity & Base Structures - Video & Lesson Transcript | Study.com
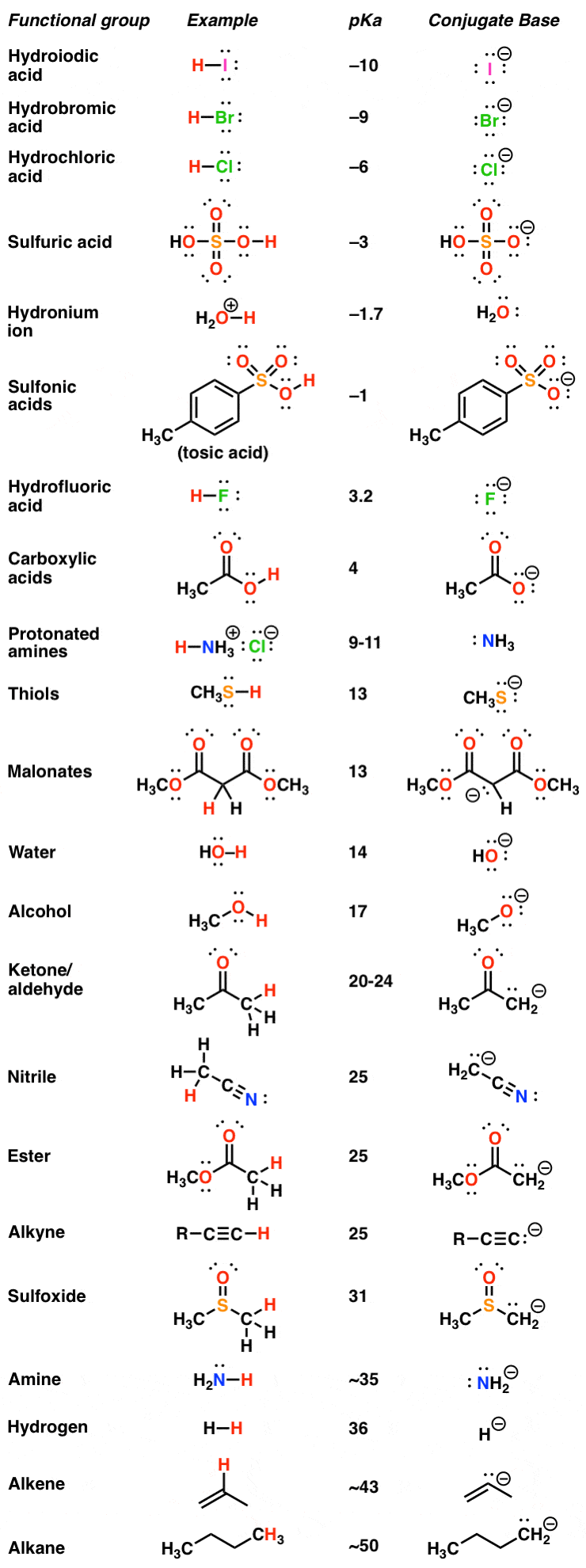
![SOLVED: PKa Table: Amino Acid PKLL COOH pK LNH Equations: FOH = 14 Kxk = 1.10 pH = Fk: Icg (A / hai) aG = AH Tas HA + 421 (Vaa * [SI]) / (+ [SII]) Pl = (Fkut pki) / 2 log[H2O] SOLVED: PKa Table: Amino Acid PKLL COOH pK LNH Equations: FOH = 14 Kxk = 1.10 pH = Fk: Icg (A / hai) aG = AH Tas HA + 421 (Vaa * [SI]) / (+ [SII]) Pl = (Fkut pki) / 2 log[H2O]](https://cdn.numerade.com/ask_images/b8cfc61f597749e79132e089dc9ece43.jpg)
SOLVED: PKa Table: Amino Acid PKLL COOH pK LNH Equations: FOH = 14 Kxk = 1.10 pH = Fk: Icg (A / hai) aG = AH Tas HA + 421 (Vaa * [SI]) / (+ [SII]) Pl = (Fkut pki) / 2 log[H2O]
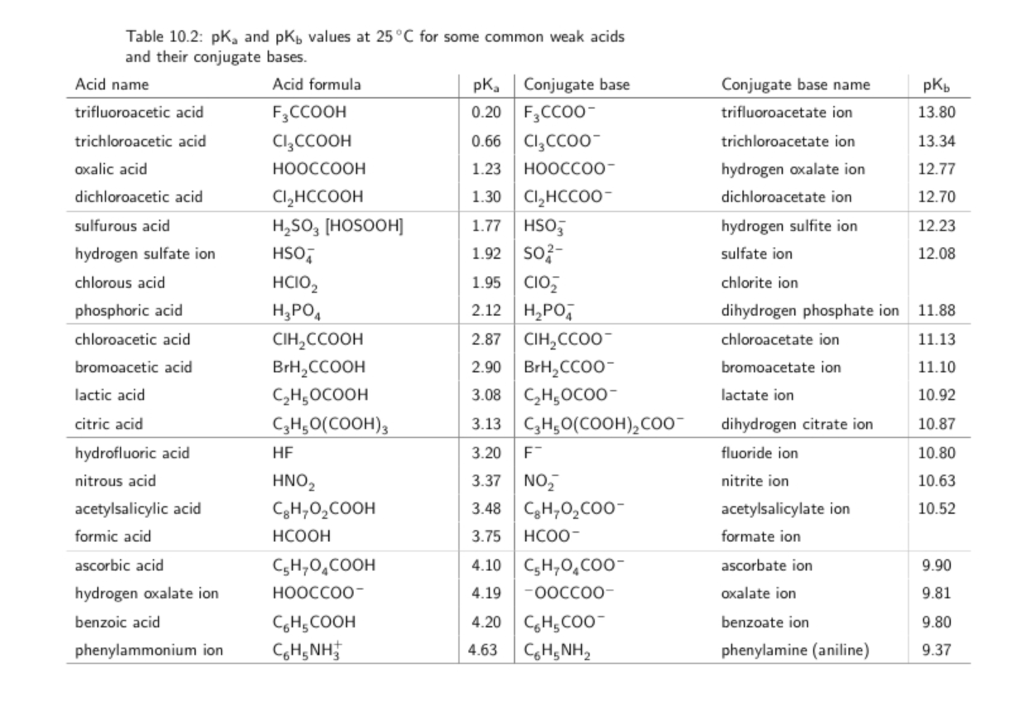
![SOLVED: The table below lists some acids and their pKa values: Acid H3PO4 2.1 CH3COOH 4.8 HPO4 7.2 HCO3- To achieve a pH around 11.0 with your selected buffer system, the [base] SOLVED: The table below lists some acids and their pKa values: Acid H3PO4 2.1 CH3COOH 4.8 HPO4 7.2 HCO3- To achieve a pH around 11.0 with your selected buffer system, the [base]](https://cdn.numerade.com/ask_images/bd3d637f77d040bf8e0900ca9ad2789b.jpg)