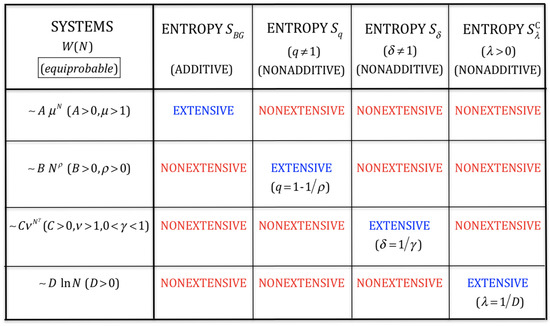
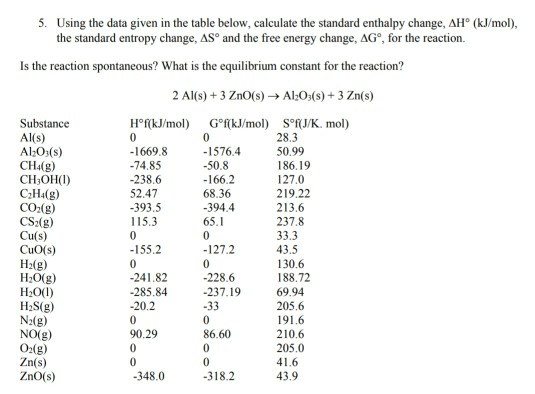
Table II from Changes in thermodynamic quantities upon contact of two solutes in solvent under isochoric and isobaric conditions. | Semantic Scholar

Table V from Large-scale calculations of gas phase thermochemistry: Enthalpy of formation, standard entropy, and heat capacity | Semantic Scholar
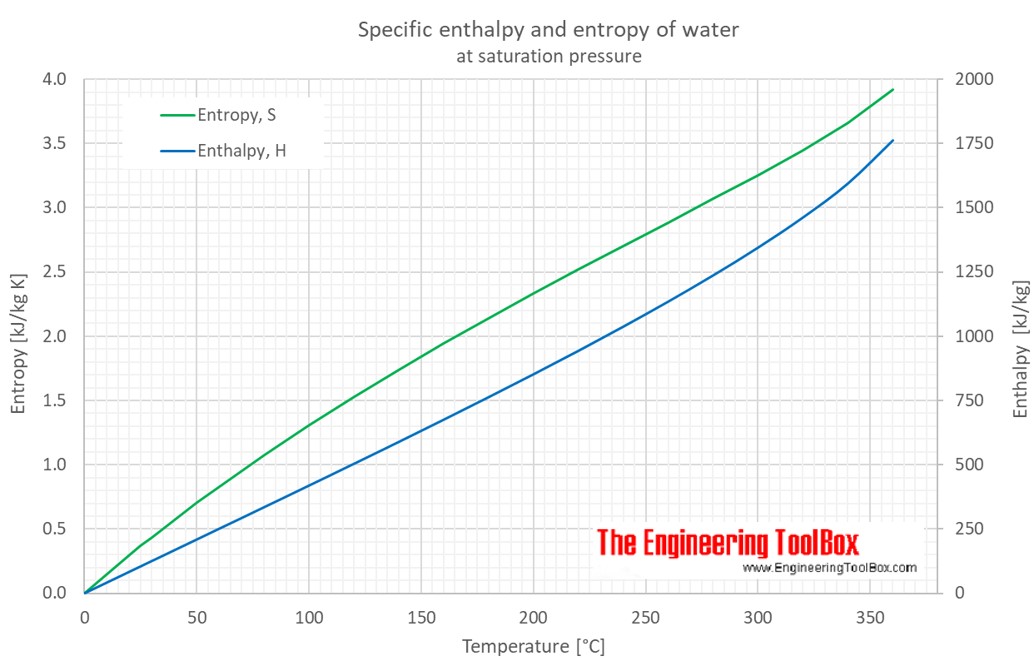
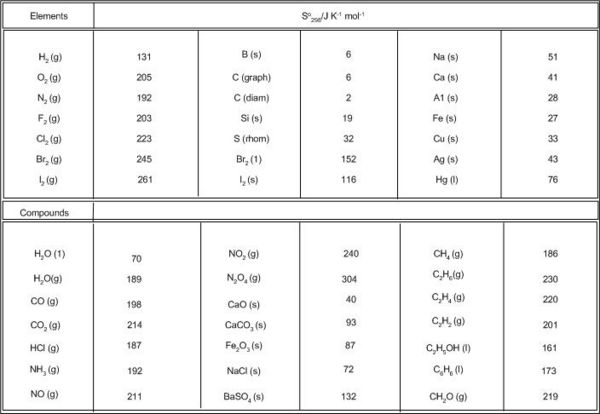
![PDF] Thermodynamics of liquids: standard molar entropies and heat capacities of common solvents from 2PT molecular dynamics. | Semantic Scholar PDF] Thermodynamics of liquids: standard molar entropies and heat capacities of common solvents from 2PT molecular dynamics. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/c3f2f7473147ea24f9502247d03ae97d7348d21a/3-Table1-1.png)
PDF] Thermodynamics of liquids: standard molar entropies and heat capacities of common solvents from 2PT molecular dynamics. | Semantic Scholar

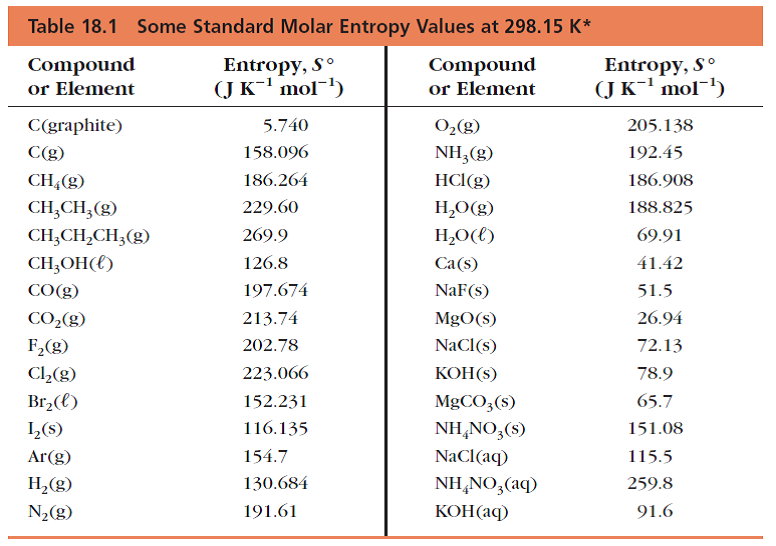
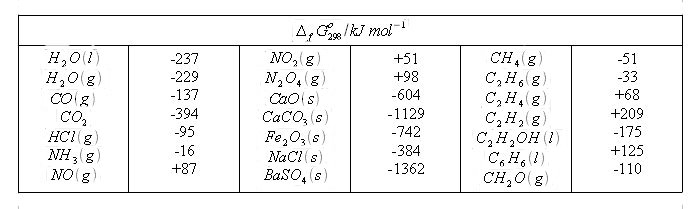
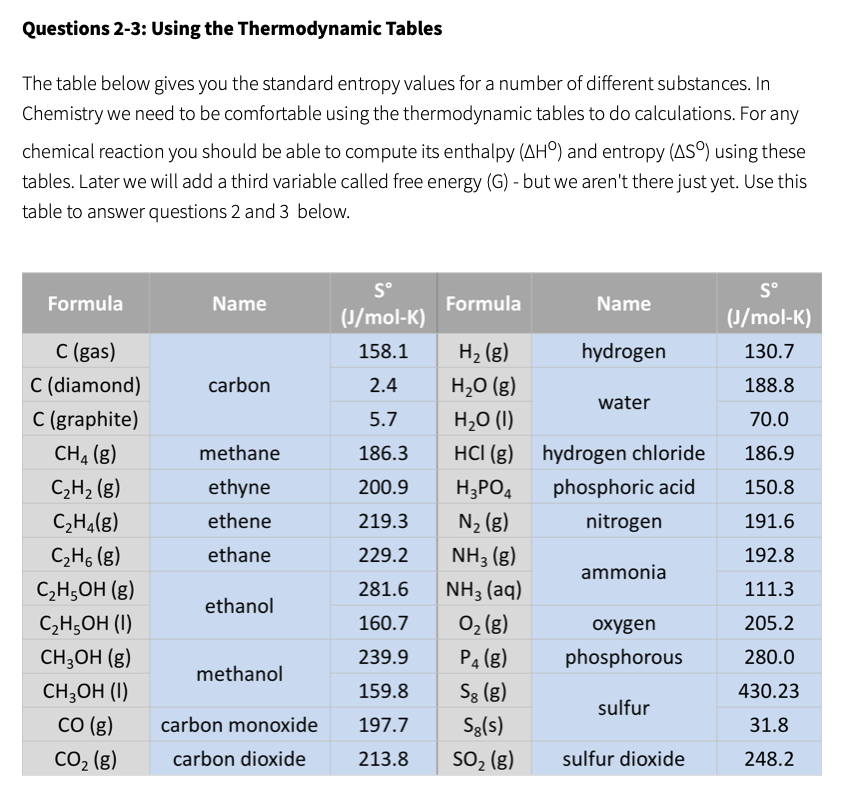
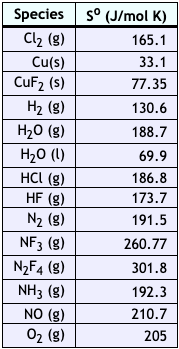
using the table of thermochemical data, calculate the change in entropy for the formation of diamond from - Brainly.com
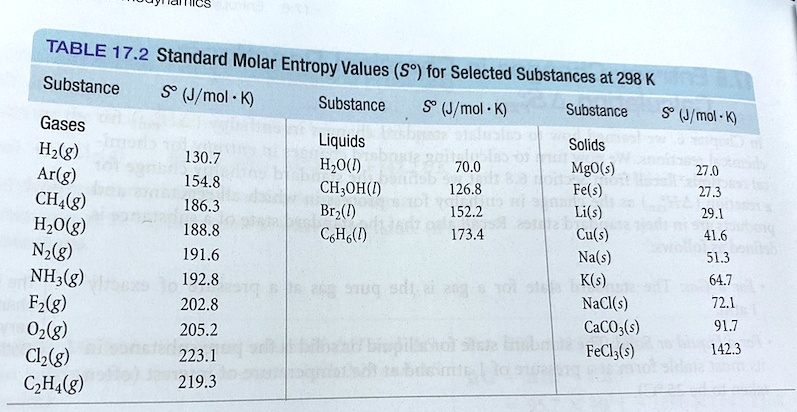
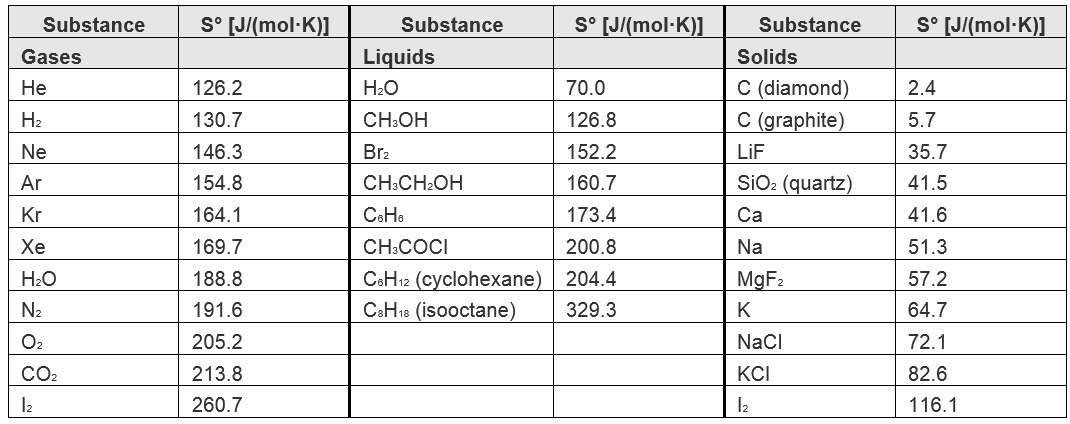
SOLVED: TABLE 17.2 Standard Molar Entropy Values (S) for Selected Substances at 298 K Substance (J/mol-K) Substance ΔS (J/mol·K) Gases H2(g) 130.7 H2O(g) 70.0 Ar(g) 154.8 CH3OH(g) 126.8 CH4(g) 186.3 Br2(g) 152.2