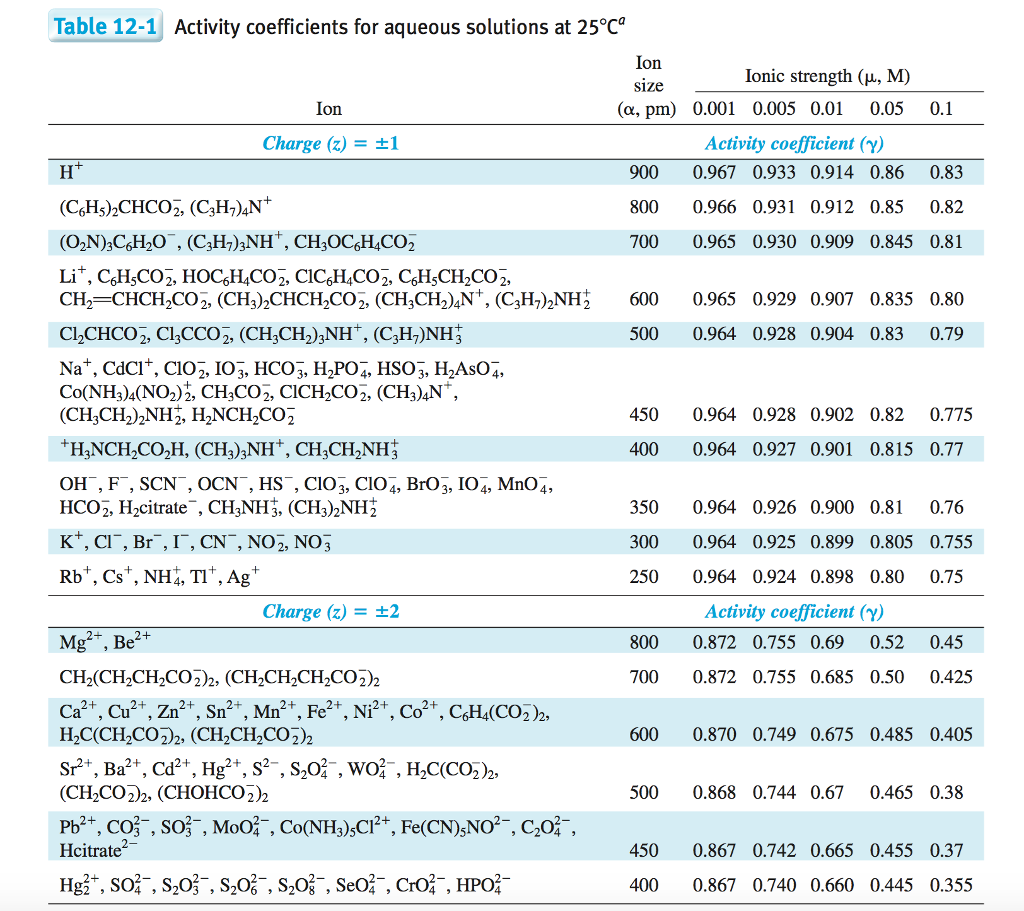
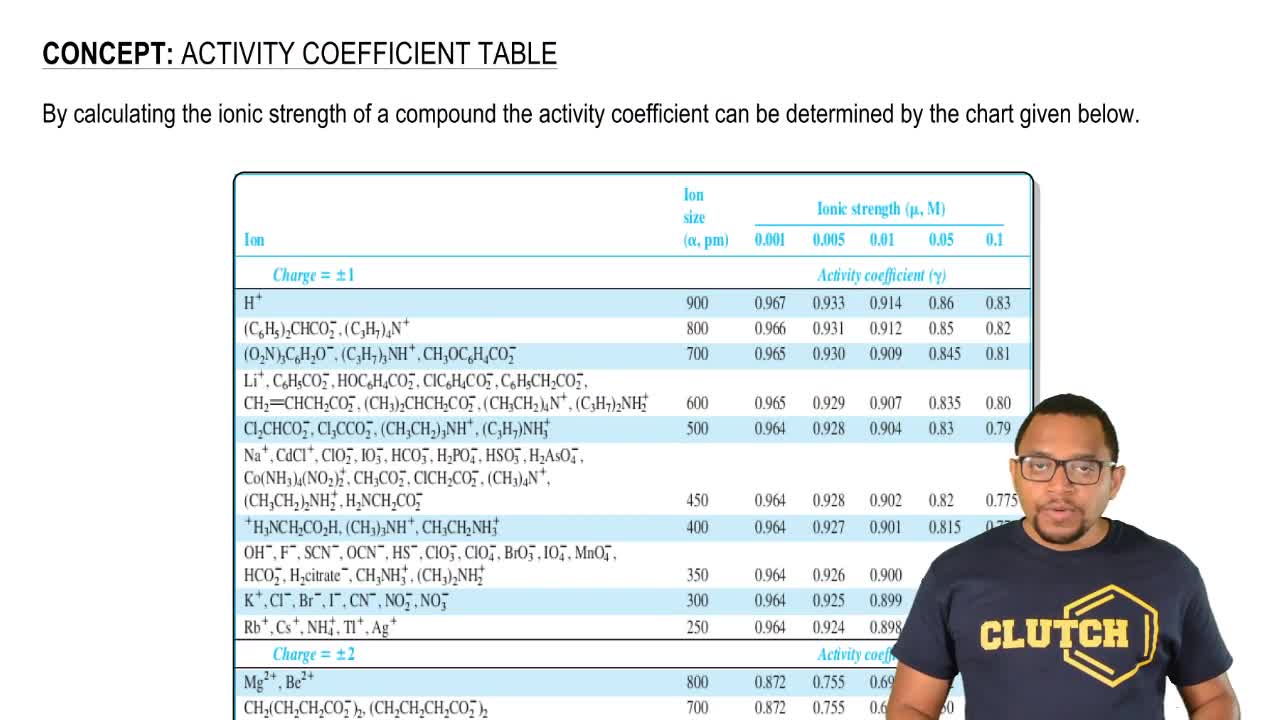
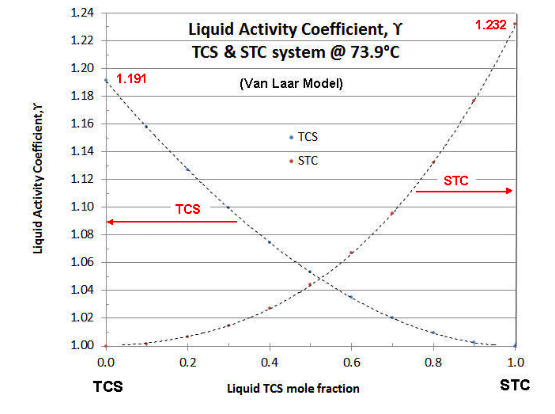
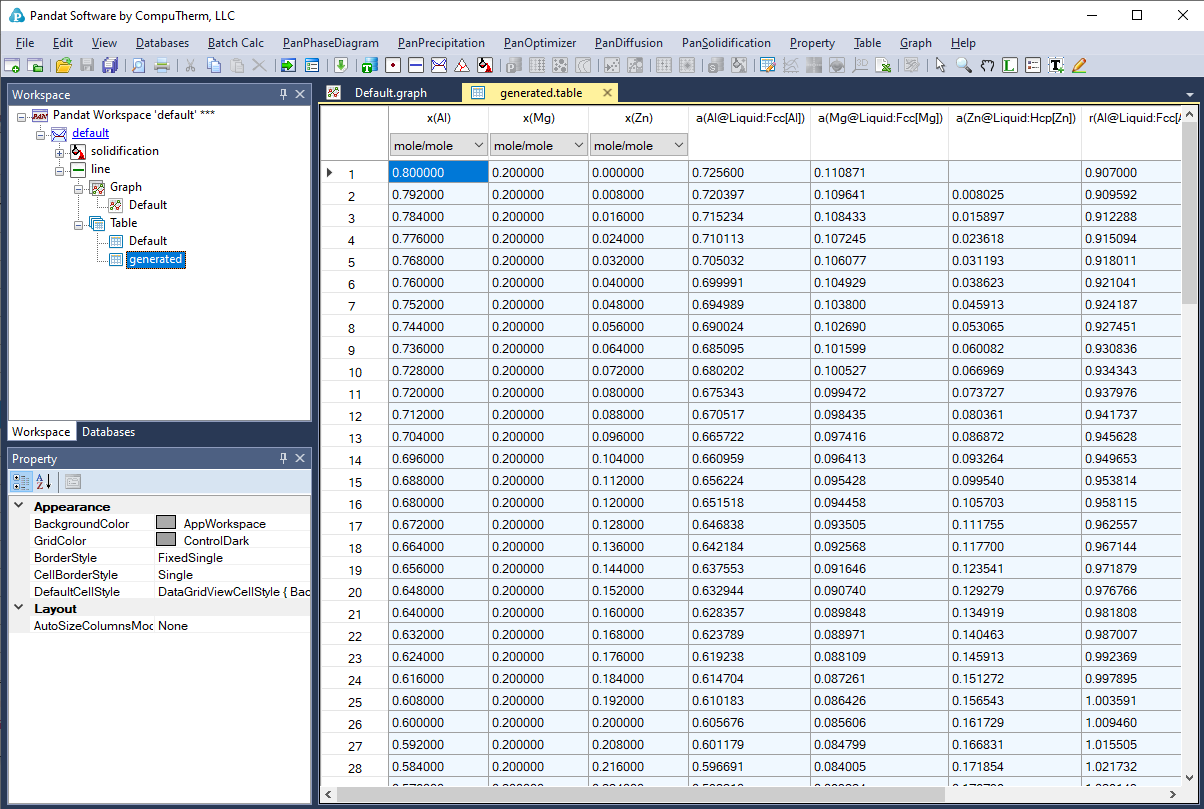
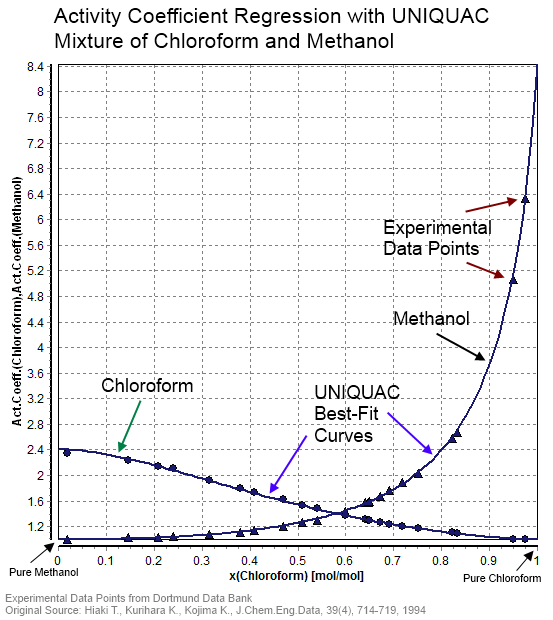
SOLVED: The table shows the value of the activity coefficient of Lit at different values of ionic strength. Interpolate the values in the table to find the activity coefficient of Lit at
![PDF] Modeling of Activity Coefficients of Aqueous Solutions of Quaternary Ammonium Salts with the Electrolyte-NRTL Equation | Semantic Scholar PDF] Modeling of Activity Coefficients of Aqueous Solutions of Quaternary Ammonium Salts with the Electrolyte-NRTL Equation | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/c6ae120eca904c5236aa29b84a028d18545bb496/41-Table2-1.png)
PDF] Modeling of Activity Coefficients of Aqueous Solutions of Quaternary Ammonium Salts with the Electrolyte-NRTL Equation | Semantic Scholar
![PDF] Measurement of Activity Coefficient at Infinite Dilution of Hydrocarbons in Sulfolane Using Gas−Liquid Chromatography | Semantic Scholar PDF] Measurement of Activity Coefficient at Infinite Dilution of Hydrocarbons in Sulfolane Using Gas−Liquid Chromatography | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/5af153d0d64d79f5da06e82e7522fa317a7e5cc1/1-Table1-1.png)
PDF] Measurement of Activity Coefficient at Infinite Dilution of Hydrocarbons in Sulfolane Using Gas−Liquid Chromatography | Semantic Scholar

1 CHAPTER 2 ELECTROLYTE SOLUTION 2-1 Strong and Weak Electrolyte Solution 2-2 Theory of Acid-base 2-3 Acidity and Calculation of Solution 2-4 Equilibrium. - ppt download

CHAPTER 5 ELECTROLYTE EFFECTS AND EQUILIBRIUM: CALCULATIONS IN COMPLEX SYSTEMS Introduction to Analytical Chemistry ppt download
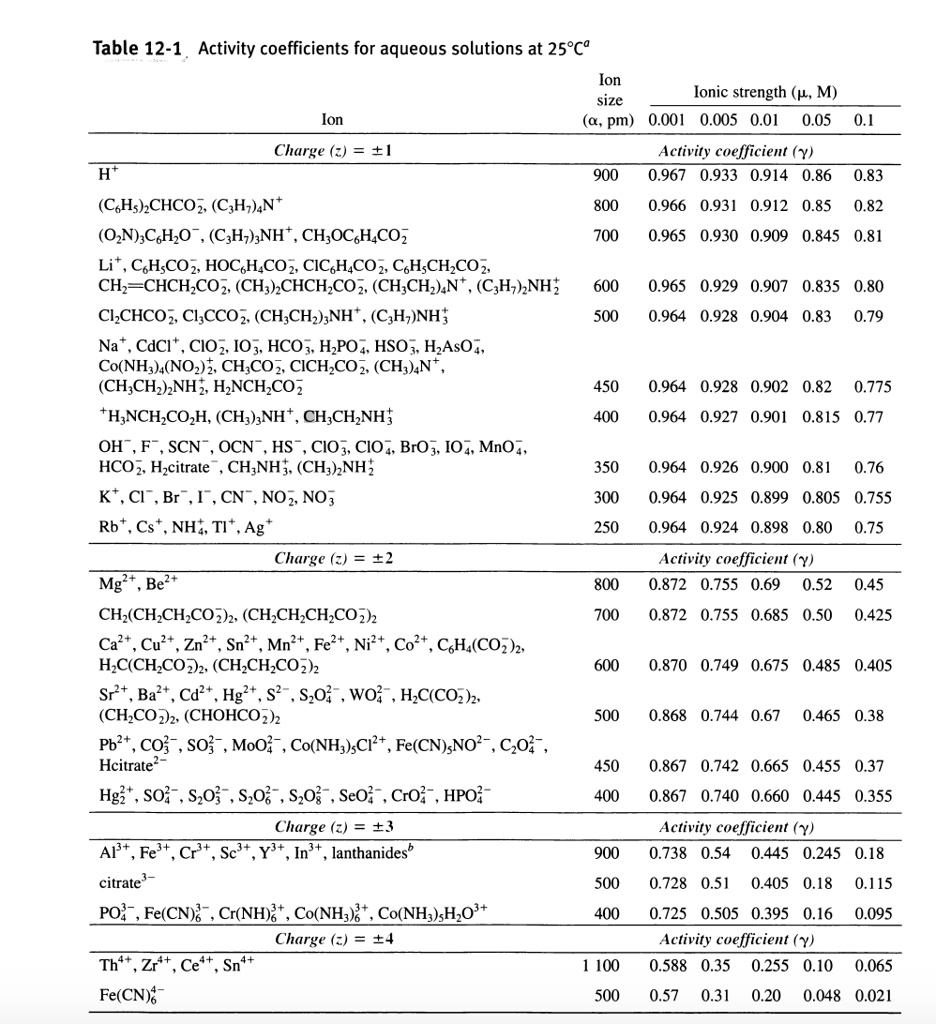
SOLVED: Table 12-1 , Activity coefficients for aqueous solutions at 25*c9 Ion Ionic strength (p, M) size Ion pm) 0.001 0.005 0.01 0.05 Charge (2) = +1 Activity cvefficient (Y) 0.967 0.933
![PDF] Activity Coefficient Models for Calculations of Supercritical CO 2 and H2O at High Temperatures and Pressures | Semantic Scholar PDF] Activity Coefficient Models for Calculations of Supercritical CO 2 and H2O at High Temperatures and Pressures | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/92bf75136f5b4d828c31a029ebf3775c09148719/4-Table1-1.png)